 This system is contraindicated for patients who are unable to operate the system or who have failed to receive effective pain relief during trial stimulation. Indications For UseĪbbott Medical spinal cord stimulation (SCS) systems are indicated as an aid in the management of chronic, intractable pain of the trunk and/or limbs, including unilateral or bilateral pain associated with the following: failed back surgery syndrome, nonsurgical back pain (without prior surgery and not a candidate for back surgery), and diabetic peripheral neuropathy of the lower extremities. The system is intended to be used with leads and associated extensions that are compatible with the system. This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. Spinal Column Stimulation (SCS) Systems Intended Use 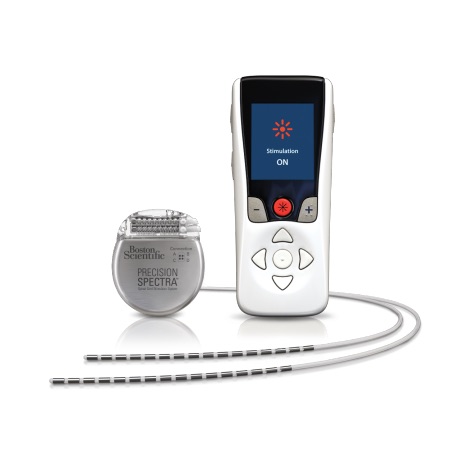
Microvascular decompression and radiofrequency for the treatment of trigeminal neuralgia: a meta-analysis. Radiofrequency denervation for treatment of sacroiliac joint pain-comparison of two different ablation techniques. Facet (Zygapophyseal) Intraarticular Joint Injections: Cervical, Lumbar, and Thoracic. Cervical zygapophysial (facet) joint pain: effectiveness of interventional management strategies. Manchikanti L, Hirsch JA, Kaye AD, Boswell MV.A systematic review and best evidence synthesis of effectiveness of therapeutic facet joint interventions in managing chronic spinal pain. Manchikanti L, Kaye AD, Boswell MV, et al.Proclaim™ DRG Neurostimulation System Clinician’s Manual. Dorsal root ganglion stimulation yielded higher treatment success rate for complex regional pain syndrome and causalgia at 3 and 12 months: a randomized comparative trial. Clinical Summaries: Spinal Cord Stimulation Systems. Spinal cord stimulation in patients with painful diabetic neuropathy: a multicentre randomized clinical trial. de Vos CC, Meier K, Zaalberg PB, et al.Spinal cord stimulation and pain relief in painful diabetic peripheral neuropathy: a prospective two-center randomized controlled trial. Slangen R, Schaper NC, Faber CG, et al.Current strategies for the management of painful diabetic neuropathy. Staudt MD, Prabhala T, Sheldon BL, et al.Pocket pain, does location matter: a single-centre retrospective study of patients implanted with a spinal cord stimulator. Baranidharan G, Bretherton B, Richert G, et al.Eterna SCS IPG Size Comparison Memo (MAT-2210151) 2022. Eterna Lowest Recharge Burden Comparison Memo (MAT 2210739) 2023. 
Eterna IPG Elect Design Verification Report: Current Draw (90860050). Eterna IPG Battery Recharge Characterization Report (90903492) 2023. Poster presented at the biannual meeting of the International Neuromodulation Society, London, England. A prospective clinical evaluation of a rechargeable implantable pulse generator (IPG): Final analysis of the sustainability of spinal cord stimulation therapy for chronic lower back pain. Randomized, controlled trial assessing burst stimulation for chronic pain: 2-year outcomes from the SUNBURST study. Success Using Neuromodulation With BURST (SUNBURST) Study: Results From a Prospective, Randomized Controlled Trial Using a Novel Burst Waveform. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
